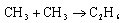
The problems below are based on the lecture course on Solution Kinetics. There is also an Oxford Primer Series by Cox, Modern Liquid Phase Kinetics.
Where the question number is enclosed in a button, e.g. , you can obtain help or comments about the question by clicking it.
1. (a) Estimate the collision frequency of molecules (of RMM 18) in (i) a gas at atmospheric pressure and 300 K, and (ii) a gas with the same density as liquid water at 300 K. Use a collision diameter of 5 Å.
(b) Estimate the diffusion controlled rate constants for the reaction of two neutral particles in (i) hexane (ii) water at 300 K. Comment on the following approximate rate constants for the recombination of solvated electrons and cations in hexane (~ 1014 dm3 mol-1 s-1) and water (~ 1010 dm3 mol -1 s-1. The viscosities of hexane and water are 3.26 x 10-4 and 1.00 x 10 -3 kg m-1 s-1 respectively.
The observed activation energy for the radical recombination process
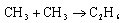
in solution is approximately 10-15 kJ mol-1. In the gas phase, the activation energy is close to zero. Comment. Explain why the reaction in solution is four times slower than might be expected on the basis of solvent viscosity.
2. In solution the mechanism for a bimolecular reaction between species A and B may be written
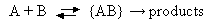
What is meant by the species {AB} appearing in this mechanism?
Derive an expression for the rate constant of a reaction in solution in terms of the elementary processes shown above. Hence explain under what conditions such a reaction would be expected to be diffusion-controlled.
The diffusion-controlled rate constant for a bimolecular reaction between species A and B in solution (expressed in dm3mol-1s-1) can be written as kD = 4000πrABDABNAf(U) where rAB is the encounter distance (expressed in m), DAB = DA + DB is the relative diffusion coefficient (in m2s-1) and f(U) is some function of the interaction potential energy, U, between the reactants, evaluated at the encounter distance. For reactions between neutral species, f(U) can be set to unity.
(a) A typical diffusion coefficient for a small molecule in aqueous solution is DA = 2.0 × 10-9m2s-1. Use this value to estimate the diffusion-controlled rate constant for a reaction involving neutral species, assuming an encounter distance of 0.5 nm.
(b) Diffusion-controlled rate constants typically decrease with increasing solvent viscosity and often have activation barriers (typically between 10 kJ mol-1 and 20 kJ mol-1). Comment on this observation.
For a reaction between ions in solution
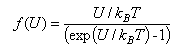
where the Coulomb potential can be written as
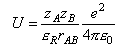
where zA and zB are the reactant charge numbers, εR is the relative permittivity, and e2/4πε0 = 2.307 × 10-28 J m.
(c) Sketch the variation of f(U) with U/kBT, and use it to explain qualitatively the effect charge has on the diffusion rate constant. Note that ex = (1 + x +..) for small x.
(d) Calculate f(U) for the reaction between solvated electrons and singly charged cations in hexane and in water at 300 K, given the relative permittivities of hexane and water are 1.89 and 78.5 respectively, and assuming an encounter distance of 0.5 nm. Comment on the result you obtain.
3. Comment on the following:
(a) For a number of ion-ion reactions in solution there exists a correlation between the entropy of activation, the volume of activation and the dependence of the reaction rate on pressure.
(b) For a series of electron transfer reactions it is sometimes possible to measure rate constants that decrease as the Gibbs energy of the reaction becomes more negative.
(c) Addition of an inert electrolyte accelerates some ion-ion reactions and decelerates others.
(d) In acid catalysed reactions addition of an inert electrolyte sometimes causes an increase in the rate of reaction.
(e) Estimate the encounter distance for the reaction of H+ and OH- in water at 300 K from the following data: diffusion coefficients, D(H+) = 9.1 x 10 -9 m2 s -1 , D(OH-) = 5.2 x 10 -9 m2 s-1 ; rate constant = 1.4 x 1011 dm3 mol -1 s-1. Comment on your answer.
4. Discuss the data given below for conductivity measurements in water, pure sulphuric acid and liquid ammonia,
| - | H2O | H2SO4 | NH3 |
| viscosity/ g m-1 s-1 | 0.29 | 25 | 0.25 |
| conductivity/ohm-1 cm2 mol-1 | - | - | - |
| Na+ | 50 | 3 | 158 |
| K+ | 74 | 5 | 176 |
| HSO4- | 52 | 151 | - |
| H+ | 350 | 152 | 142 |